J.A. Isaac, G. Gellon, F. Molton, C. Philouze, N. Le Poul, C. Belle, A. Thibon-Pourret. Symmetrical and unsymmetrical dicopper complexes based on bis-oxazoline units: synthesis, spectroscopic properties and reactivity. Inorganics 2023, 11, 332-347. DOI: 10.3390/inorganics11080332
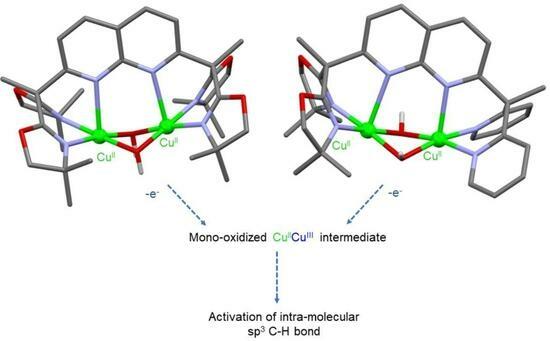
Copper–oxygen adducts are known for being key active species for the oxidation of C–H bonds in copper enzymes and their synthetic models. In this work, the synthesis and spectroscopic characterizations of such intermediates using dinucleating ligands based on a 1,8 naphthyridine spacer with oxazolines or mixed pyridine-oxazoline coordination moieties as binding pockets for copper ions have been explored. On the one hand, the reaction of dicopper(I) complexes with O2 at low temperature led to the formation of a μ-η2:η2 Cu2:O2 peroxido species according to UV-Vis spectroscopy monitoring. The reaction of these species with 2,4-di-tert-butyl-phenolate resulted in the formation of the C–C coupling product, but no insertion of oxygen occurred. On the other hand, the synthesis of dinuclear Cu(II) bis-μ-hydroxido complexes based on pyridine–oxazoline and oxazoline ligands were carried out to further generate CuIICuIII oxygen species. For both complexes, a reversible monoelectronic oxidation was detected via cyclic voltammetry at E1/2 = 1.27 and 1.09 V vs. Fc+/Fc, respectively. Electron paramagnetic resonance spectroscopy (EPR) and UV-Vis spectroelectrochemical methods indicated the formation of a mixed-valent CuIICuIII species. Although no reactivity towards exogeneous substrates (toluene) could be observed, the CuIICuIII complexes were shown to be able to perform hydroxylation on the methyl group of the oxazoline moieties. The present study therefore indicates that the electrochemically generated CuIICuIII species described herein are capable of intramolecular aliphatic oxidation of C–H bonds.